
Image: Atomwise CEO and co-founder Abraham Heifets
Chinese review designations lag behind US and EU
GlobalData explores why Chinese review designations currently lag behind the US and the EU in terms of the number of designations awarded.
Dr Judith M. Sills. Credit: Arriello
Dr Eric Caugant. Credit: Arriello
The introduction of review designations by China’s National Medical Products Administration (NMPA) in 2015 has shown some success, with more than 190 drugs awarded a priority review, breakthrough therapy or accelerated approval review designation. This has allowed China to overtake Japan, but the country still lags behind the US and EU markets in terms of the number of drug designations awarded.
China reformed its regulatory system in 2015 to include various fast-track programmes to drive the development of drugs for unmet medical needs and serious conditions, launching priority review, breakthrough therapy and accelerated approval designations. The benefits of these designations include earlier approval times and faster review times by the NMPA.
For example, Shanghai Junshi Bioscience’s toripalimab, a monoclonal antibody indicated for metastatic melanoma, was awarded the acceperated approval designation in March 2018. Toripalimab was the first domestic programmed cell death protein 1 (PD-1) monoclonal antibody approved in China.
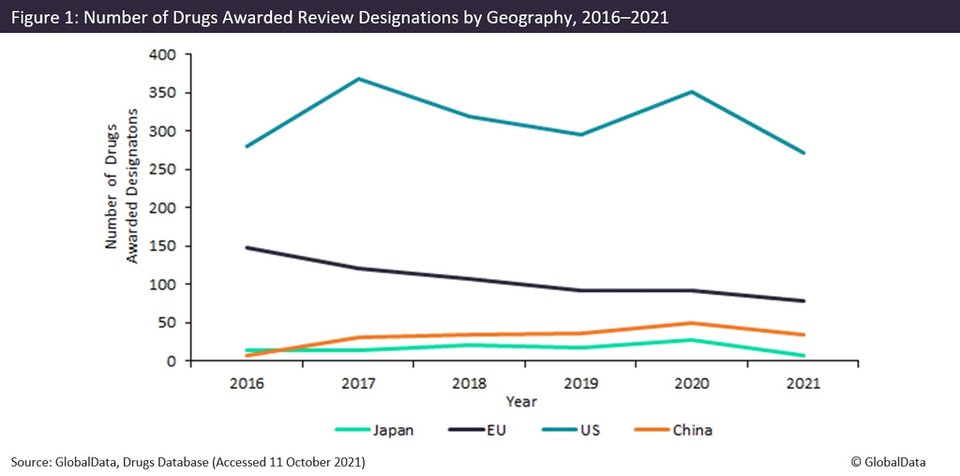
According to GlobalData’s Pharma Intelligence Centre Drugs database, the NMPA awarded review designations to 108% more drug candidates per year than Japan from 2017 to this October. Japan introduced its own review designation scheme in 2017, named Sakigake, as part of the wider Japan Revitalisation Strategy intended to expedite innovative pharmaceuticals’ path to market and to promote the involvement of foreign companies.
China, however, remains behind the European Medicines Agency and US Food and Drug Administration in terms of the number of drugs awarded review designations. As shown in figure 1, the US awarded around nine times more designations than China and the EU awarded three times more designations than China over the same six-year time period.
The US and EU review designation programmes were, however, implemented more than 20 years ago; the US awarded its first designation as an orphan drug designation in 1983, with the EU following suit in 2000.
China seeks to continue to drive domestic development with regulatory changes and novel approval pathways to drive growth in innovator therapeutics and unmet needs. While Chinese review designations currently lag behind the US and the EU designation award numbers, it is likely that going forward, it will aim to increase the number of drugs awarded designations to match these industry leaders.
COMMENT from