Validated, Sleek, Clinical Kit Temperature Datalogger
Validated, sleek, USB
temperature datalogger fully
validated for mission-critical
clinical trials.
USB temperature monitor that is fully validated for mission-critical clinical trials
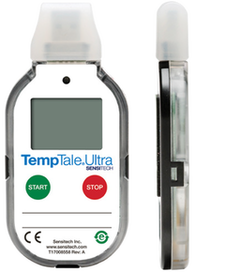
Every clinical trial is unique. Minimize risk to clinical trial materials by monitoring shipments in compliance with GDP and FDA regulations. The TempTale Ultra datalogger is the intuitive next-generation of world-class TempTale products. It can be tailored to support up to six programmable time and temperature alarms, including, Mean Kinetic Temperature (MKT) and duration per clinical protocols.
TempTale Ultra Highlights
- Thin 12.5mm housing – easily slips into clinical kits, with large LCD screen.
- PDF configurable reports support local time zones and five languages.
- Simple ColdStream Web upload for exception quality review; integrates with IVR/IRT systems.
- Program Management support available for analyzing summary shipment statistics for smart site and cost reduction recommendations.
For more information please call +31(0) 252 211 108
