DEALS ANALYSIS
Deals activity: South and Central America YoY increase; private equity deal drop
Powered by
Deals activity by geography
Pharma industry deals, as captured by GlobalData’s Pharmaceuticals Intelligence Centre, are up year-on-year (YoY) across all regions.
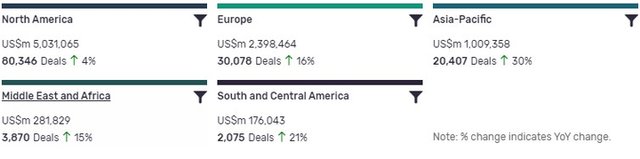
North America is leading in terms of deal value, but recorded the lowest YoY growth in deals volume at 4%. Asia-Pacific, ranking third in terms of deal value, has seen the biggest YoY change, with deal volumes increasing by 30%.
The volume of deals recorded by GlobalData also increased YoY in South and Central America (21%), Europe (16%) and Middle East and Africa (15%).
Deals activity by type
| Deal type | Total deal value (US$m) | Total deal count | YoY change (volume) |
| Partnership | 459528 | 17704 | 0 |
| Venture Financing | 211013 | 17549 | 41 |
| Contract Service Agreement | 1849 | 16956 | 665 |
| Equity Offering | 851620 | 15788 | 142 |
| Licensing Agreement | 679040 | 13308 | 52 |
| Grant | 6529 | 12600 | -34 |
| Acquisition | 3593731 | 10564 | -46 |
| Debt Offering | 1753561 | 7809 | 28 |
| Asset Transaction | 424069 | 6011 | -54 |
| Private Equity | 498132 | 244 | -22 |
| Merger | 167235 | 660 | 283 |
A breakdown of deals by type and volume shows a 283% growth in mergers YoY, while acquisitions are down 46%, partnerships held steady with 0% change YoY and asset transactions are down -54%. Financing deals have increased across some types, with venture financing up 41% YoY, equity offerings up 142% and debt offerings up 28%.
Private equity, however, has seen a drop of -22% in number of deals YoY, while the number of grants recorded is down -34%. The number of contract service agreements recorded by GlobalData is up 665% year-on-year.
Deals activity by therapy area
The most notable development apparent in GlobalData’s analysis of pharma industry deals by therapy area is the increase of deals in the field of infectious diseases. After remaining relatively steady over the past ten years, the number of recorded deals in infectious diseases increased significantly in 2020 to date, most likely due to the impact of the Covid-19 pandemic on activity in the sector.
Note: All numbers as of 16 November 2020. Deals captured by GlobalData cover M&As, strategic alliances, various types of financing and contract service agreements.
For more insight and data, visit GlobalData's Pharmaceuticals Intelligence Centre.
Latest deals in brief
Taiho signs drug licensing deal with Lung Therapeutics in Japan
Taiho Pharmaceutical has entered an exclusive licence agreement with Lung Therapeutics for the latter’s investigational recombinant human single-chain urokinase plasminogen activator, LTI-01, in Japan. According to the deal, Taiho will gain the rights to develop and commercialise LTI-01 in the country. Lung Tx will get an upfront payment, milestone and royalty payments depending on the product sales.
IMCR and SII collaborate to develop Novavax’s Covid-19 vaccine in India
Serum Institute of India (SII) and Indian Council of Medical Research have collaborated for clinical development of the Covid-19 vaccine, COVOVAX, by US-based firm Novavax and upscaled by SII. Novavax has also initiated its late-phase trials of the vaccine in South Africa and the UK and plans to start the same in the US soon. For the production of the vaccine, SII has received the bulk vaccine and Matrix-M adjuvant from the US firm and will fill and finish them in vials.
Pfizer and BioNTech sign Covid-19 vaccine supply deal with European Commission
Pfizer and BioNTech have reached an agreement with the European Commission (EC) for the supply of 200 million doses of their investigational BNT162b2 mRNA-based vaccine candidate against the SARS-CoV-2 virus that causes Covid-19. In September, the companies concluded exploratory talks with the EC to supply doses of the vaccine candidate, if approved, and the latest supply agreement is now final. Under the agreement, Pfizer and BioNTech will supply BNT162b2 to European Union Member States.
Bridge Biotherapeutics terminates collaboration agreement with Boehringer
Bridge Biotherapeutics and Boehringer Ingelheim have mutually agreed to terminate their previously signed collaboration and licence agreement to develop BBT-877. According to the deal, Boehringer agreed to make upfront and near-term payments of €45m to Bridge Biotherapeutics. This deal included milestone payments of up to €1.1bn.
Humanigen signs Covid-19 therapy development deal with US
Humanigen has signed an agreement with the US Department of Defense Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense to develop lenzilumab for Covid-19 treatment. The Cooperative Research and Development Agreement, signed in partnership with the Biomedical Advanced Research and Development Authority aims to aid the government’s Operation Warp Speed programme.
Merck to acquire oncology firm VelosBio for $2.75bn
Merck has signed a definitive agreement to acquire all outstanding shares of Pappas Capital’s portfolio company VelosBio through a subsidiary for $2.75bn in cash, subject to certain customary adjustments. Pioneer in cancer therapies that target receptor tyrosine kinase-like orphan receptor 1, VelosBio is developing VLS-101, an investigational ADC comprising a monoclonal antibody. The acquisition will strengthen Merck’s oncology pipeline with the addition of VelosBio’s lead investigational candidate VLS-101.